 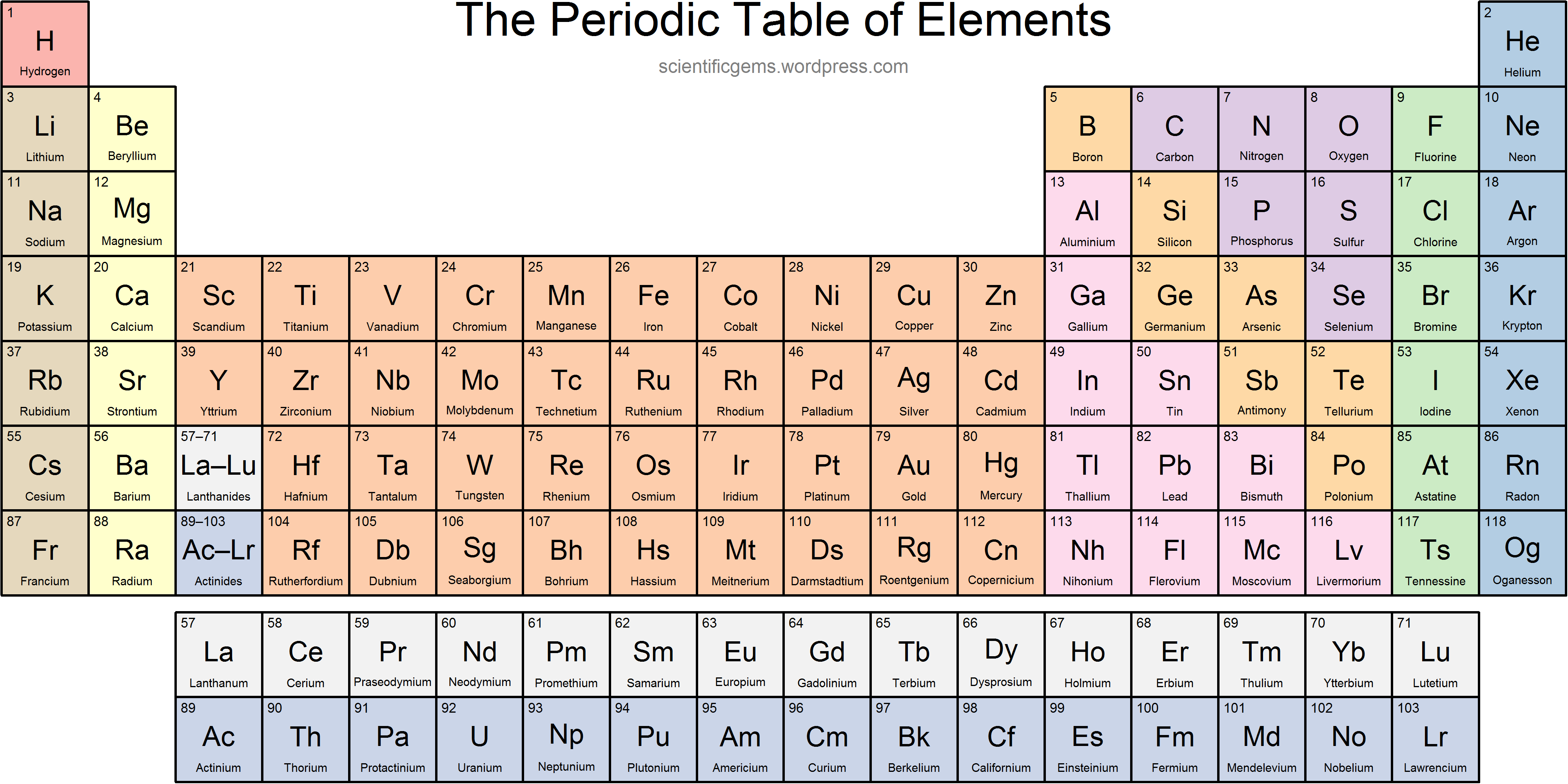 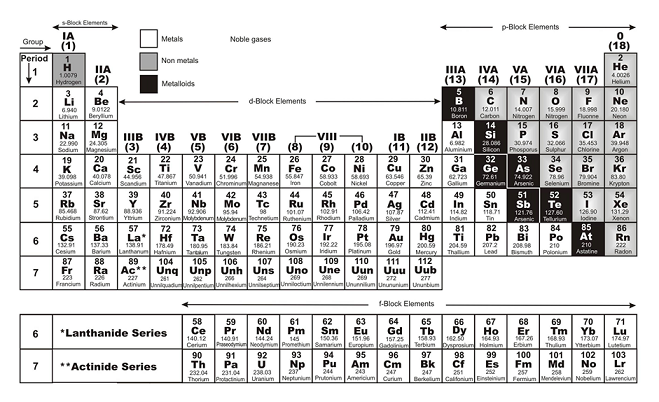
Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table containing elements with partially filled p orbitals. The nonmetals are located on the upper right side of the periodic table. Nonmetals include solids, liquids, and gases at room temperature and pressure.They tend not to be malleable or ductile, so they form brittle solids. Traditionally, in the United States the taller. Nonmetals are typically poor conductors of heat and electricity, with low melting and boiling points. Periods: the horizontal rows on the table, with the elements arranged in order of increasing atomic number.For example, diamond and graphite are both pure carbon. As you go along periods the number of protons and neutrons increases and the number of electron shells remains the. There are seven periods total and each element in a period has the same number of atomic orbitals. Periods are the rows of the periodic table. For the transition metals with partially-filed d shells, valence electrons are those electrons outside the noble gas core. Periods are the horizontal rows of the periodic table. He organized the elements by atomic number, which is. 
Valence electrons are outer shell electrons for main group elements. The periodic table of the elements was first introduced in the mid-19th century by Dmitry Mendeleev. Most nonmetals take many forms called allotropes. In chemistry and physics, a valence electron is an electron associated with an atom that can form a chemical bond and participate in a chemical reactions.In each period the left most element has 1 electron in. Nonmetals typically don't look metallic. The first one is short and only has two elements, hydrogen and helium.The nonmetals include the group called the nonmetals, as well as the halogens and the noble gases.The number of valence electrons depends on the octet rule. Element Groups Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. For example, magnesium (atomic weight 24.3) is placed to the right of sodium (atomic weight 23.0): In 1913, chemistry and physics were topsy-turvy. A higher atomic weight than the one on its left. These elements are located on the righthand side of the table, with the exception of hydrogen. An element period is a horizontal row on the periodic table. The periodic table we use today is based on the one devised and published by Dmitri Mendeleev in 1869.The nonmetals are a small group of elements on the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
